Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
G M N BASTON*; J A BERRY*; M BROWNSWORD*; D J LLETT*; C M LINKLATER*; S W SWANTON*; Tweed, C. J.*
JNC TJ8400 99-078, 72 Pages, 1999/03
A desk study has been carried out to establish the feasibility of measuring the oxidation state of plutonium under near-neutral strongly-reducing conditions. X-ray absorbance spectroscopy appears to be capable of establishing the oxidation state of plutonium sorbed on a suitable substrate. An experimental and modelling investigation has been performed to study the sorption of plutonium onto basalt, mudstone and sandstone under strongly-reducing conditions at three concentrations of carbonate. Appropriate synthetic rock-equilibrated de-ionised water and seawater were used. A model has been developed to describe the sorption of plutonium onto basalt, mudstone and sandstone in de-ionised water and seawater. Predicted R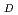 values are generally in good agreement with the observed experimental measurements. The model is based on sorption of plutonium(III) species and assumes iron oxide is the dominant sorbing phase.
values are generally in good agreement with the observed experimental measurements. The model is based on sorption of plutonium(III) species and assumes iron oxide is the dominant sorbing phase.
 and spent fuel; A Review
and spent fuel; A ReviewKitamura, Akira; Akahori, Kuniaki*
no journal, ,
Since dissolution rate of UO matrices will be depend on carbonate concentration due to promoting oxidative dissolution of spent nuclear fuel by formation of carbonate complexes of uranium(VI), effect of carbonate concentration on dissolution rate of UO
matrices will be depend on carbonate concentration due to promoting oxidative dissolution of spent nuclear fuel by formation of carbonate complexes of uranium(VI), effect of carbonate concentration on dissolution rate of UO and spent nuclear fuel has been reviewed. It is found that a systematic study on dissolution rate of UO
and spent nuclear fuel has been reviewed. It is found that a systematic study on dissolution rate of UO and/or spent fuel as a function of carbonate concentration is recommended.
and/or spent fuel as a function of carbonate concentration is recommended.